Introduction: The Silent Incubation
For decades, the public has viewed heart disease as a “late-life accident”—a sudden collision of age and lifestyle that strikes in the golden years. However, as a preventive health scientist, I can tell you that the modern medical paradigm has shifted: atherosclerotic cardiovascular disease (ASCVD) is a life-course metabolic progression that begins decades before the first symptom appears.
While clinical events like heart attacks emerge in middle age, the silent incubation of these conditions spans nearly a century. The biological clock doesn’t start at fifty; it starts in the womb. To understand heart health, we must investigate the cradle, where the foundations of cardiovascular pathology are established during the most formative stages of human development.
The 16-Month Milestone: Adult Levels in Diapers
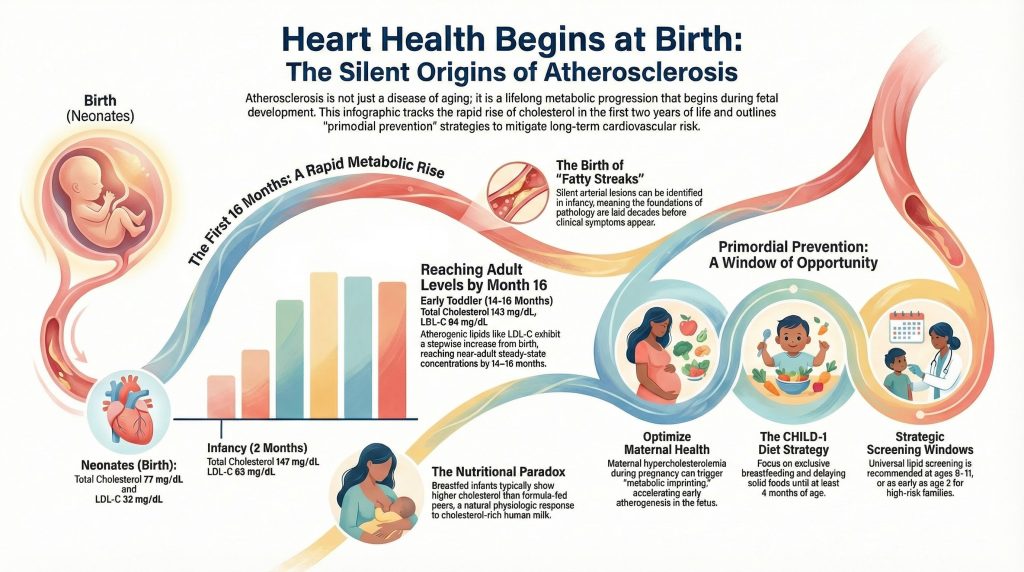
The neonatal period is a phase of radical metabolic transition. At birth, cholesterol concentrations in umbilical cord blood are significantly lower than adult levels, but this “clean slate” is short-lived. Data from the Copenhagen Baby Heart Study, which analyzed over 13,000 samples, reveals a rapid, stepwise increase in atherogenic lipids like LDL-C and apolipoprotein B (apo B) immediately after birth.
Remarkably, by 14–16 months of age—and potentially as early as 6 months—these lipid concentrations reach a steady state that is statistically indistinguishable from adult maternal levels. This “adult baseline” is established while the child is still in diapers. Intriguingly, female infants generally exhibit significantly higher concentrations of total cholesterol, LDL-C, and apo B than their male counterparts from birth, a disparity that persists through the first two years.
Perhaps most critical for parents is the early footprint of lipoprotein(a) [Lp(a)]. If an infant’s Lp(a) levels are above the 90th percentile at birth, it serves as a robust predictor that they will reach adult-range high-risk concentrations as early as 14 months of age. This highlights a counter-intuitive reality: the metabolic trajectory of a toddler can mirror the long-term risk profile of an adult before they have even mastered walking. 

Fetal Programming: The Aorta’s First Lessons
The cardiovascular journey begins before the first breath, dictated by a biological aging set-point established in utero. The maternal environment acts as the primary instructor for fetal metabolism. When a mother experiences hypercholesterolemia during pregnancy, it acts as a potent stimulus for early atherogenesis in the fetus. Findings from the FELIC study in Naples demonstrated that maternal LDL levels correlate directly with the size of atherosclerotic lesions in the fetal aorta.
This is more than a temporary exposure; it is a permanent metabolic reprogramming. As the research indicates:
“This association is likely mediated through epigenetic mechanisms… maternal cholesterol levels are positively associated with the methylation of the promoter in fetal aortas… This ‘fetal programming’ may lead to a permanent alteration in the offspring’s metabolic set-point.”
Further evidence of this biological clock is found in telomere length (TL). Shorter telomeres at birth are not just markers of cellular age; they are predictive of midlife health. A 40-year longitudinal study found that individuals born with shorter telomeres had a 3.24× increased risk of midlife hypercholesterolemia and significantly higher risk scores for arterial damage by age 42.
The Pathological Reality: Fatty Streaks in Infancy
The most jarring evidence for early intervention comes from autopsy studies. Atherosclerosis begins as fatty streaks—microscopic accumulations of lipid-engorged macrophage foam cells in the arterial wall. A Japanese nationwide study revealed that 29% of infants younger than 1 year already possessed these fatty streaks in their aortas. In some regions, such as Taiwan, pathological examinations have even shown a 17% narrowing of the left coronary artery in infants.
Early Warning Signs
| Age Group | Pathological Prevalence | Pathological Feature |
| Infants (< 1 year) | 29% (Aorta) | Sudan IV–positive fatty streaks; macrophage foam cells |
| Infants (< 1 year) | 17% (Left coronary) | Intimal proliferation; narrowing of the vessel |
| Children (Age 3) | Ubiquitous | Global presence of aortic fatty streaks |
| Adolescents (15–19) | 50% (Coronary) | Initial lipid accumulation; progressing lesions |
The Breastfeeding Paradox: When High Cholesterol is Healthy
There is a nutritional paradox in early life: breastfed infants consistently show higher total cholesterol and LDL-C than those who are formula-fed. In an adult, these numbers would trigger alarm, but in an infant, this is often a salutogenic (health-promoting) effect.
Human milk is naturally high in fat and cholesterol to support rapid myelin formation in the brain and cell membrane synthesis. This reflects the unique metabolic plasticity of infancy. As the infant transitions from a primarily lipid-based fuel source (milk) to a carbohydrate-inclusive diet, their body handles these lipids as essential building blocks rather than pathological waste. This physiological response is distinct from the chronic, dysfunctional high cholesterol seen in adults.
Vascular Memory: The Legacy of Childhood Lipids
The lipids circulating in a child’s blood leave a permanent imprint on their arteries through vascular memory. Data from the Bogalusa Heart Study and the Young Finns Study have shown that childhood LDL-C and BMI are highly predictive of carotid artery intima-media thickness (cIMT) 20 to 30 years later.
This memory means the metabolic insults of youth are stored in the arterial wall, manifesting as clinical disease in adulthood. As the Bogalusa researchers famously concluded:
“Heart disease is not an adult problem but a childhood problem with adult manifestations.”
The Modern Paradox: Better Numbers, Bigger Risks
Historical data presents a deceptive picture. Mean total cholesterol in U.S. youth has dropped from 165 mg/dL in the late 1980s to roughly 158 mg/dL today, largely due to the removal of trans fats and reduced tobacco exposure.
However, this statistical success is being cannibalized by the obesity epidemic. While the average child may look better on paper, the high-risk obesity tail of the population is larger and more vulnerable than ever. Furthermore, modern environmental stressors—specifically chronic sleep deprivation—act as physiological disruptors. Sleep loss is not merely a lifestyle issue; it triggers a cascade of hunger hormones and inflammation that acts as a catalyst for metabolic syndrome, potentially neutralizing decades of dietary progress.
The PDAY Score: Calculating the Biological Clock
The Pathobiological Determinants of Atherosclerosis in Youth (PDAY) study allows us to quantify this early damage. By correlating antemortem risk factors with arterial findings in young people who died of external causes, researchers developed a risk score that predicts future calcification with high accuracy.
The PDAY risk score is calculated using several modifiable components:
- Non-HDL and HDL cholesterol
- BMI (obesity)
- Smoking status
- Hypertension
- Hyperglycemia
The biological clock measured by this score ticks with alarming speed. In men, the prevalence of advanced Grade 4 and 5 lesions—plaques with a distinct lipid core—jumps from 2.4% at ages 15–19 to a staggering 20.3% by ages 30–34. This rapid acceleration is fueled by the cumulative exposure to risk factors that began in infancy.
Conclusion: The Window of Opportunity
We currently face a narrow window of opportunity—a brief period in early life when vascular damage is still minor and potentially reversible. Research suggests that maintaining an LDL-C level below 2 mmol/L throughout the life course makes ASCVD extremely rare. However, waiting until middle age to address these numbers is often too late, as calcified plaques may have already solidified the disease.
To change the global trajectory of heart disease, we must prioritize primordial prevention. This means optimizing maternal health and perhaps considering a provocative shift in policy: universal screening for conditions like familial hypercholesterolemia in infancy. Identifying high-risk individuals in the cradle is our best chance at ensuring the ideal cardiovascular health most babies are born with is protected for a century.
References
- Holven KB. Plasma cholesterol levels in infancy: a neglected resource. Eur Heart J. 2023;44(42):4419-4421. doi:10.1093/eurheartj/ehad558
- Taageby Nielsen S, Mohr Lytsen R, Strandkjær N, et al. Significance of lipids, lipoproteins, and apolipoproteins during the first 14-16 months of life. Eur Heart J. 2023;44(42):4408-4418. doi:10.1093/eurheartj/ehad547
- Luca AC, David SG, David AG, et al. Atherosclerosis from Newborn to Adult-Epidemiology, Pathological Aspects, and Risk Factors. Life (Basel). 2023;13(10):2056. Published 2023 Oct 14. doi:10.3390/life13102056
- Napoli C, Glass CK, Witztum JL, Deutsch R, D’Armiento FP, Palinski W. Influence of maternal hypercholesterolaemia during pregnancy on progression of early atherosclerotic lesions in childhood: Fate of Early Lesions in Children (FELIC) study. Lancet. 1999;354(9186):1234-1241. doi:10.1016/S0140-6736(99)02131-5
- de Nigris F, Cacciatore F, Mancini FP, et al. Epigenetic Hallmarks of Fetal Early Atherosclerotic Lesions in Humans. JAMA Cardiol. 2018;3(12):1184-1191. doi:10.1001/jamacardio.2018.3546
- Natural history of aortic and coronary atherosclerotic lesions in youth. Findings from the PDAY Study. Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group. Arterioscler Thromb. 1993;13(9):1291-1298. doi:10.1161/01.atv.13.9.1291
- McGill HC Jr, McMahan CA, Zieske AW, et al. Association of Coronary Heart Disease Risk Factors with microscopic qualities of coronary atherosclerosis in youth. Circulation. 2000;102(4):374-379. doi:10.1161/01.cir.102.4.374
- Strong JP, Malcom GT, McMahan CA, et al. Prevalence and extent of atherosclerosis in adolescents and young adults: implications for prevention from the Pathobiological Determinants of Atherosclerosis in Youth Study. JAMA. 1999;281(8):727-735. doi:10.1001/jama.281.8.727
- Takei H, Strong JP, Yutani C, Malcom GT. Comparison of coronary and aortic atherosclerosis in youth from Japan and the USA. Atherosclerosis. 2005;180(1):171-179. doi:10.1016/j.atherosclerosis.2004.11.014
- Françoso LA, Coates V. Anatomicopathological evidence of the beginning of atherosclerosis in infancy and adolescence. Arq Bras Cardiol. 2002;78(1):131-142.
- Øyri LKL, Bogsrud MP, Kristiansen AL, et al. Cholesterol at ages 6, 12 and 24 months: Tracking and associations with diet and maternal cholesterol in the Infant Cholesterol Study. Atherosclerosis. 2021;326:11-16. doi:10.1016/j.atherosclerosis.2021.04.017
- Gidding SS, Rana JS, Prendergast C, et al. Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Risk Score in Young Adults Predicts Coronary Artery and Abdominal Aorta Calcium in Middle Age: The CARDIA Study. Circulation. 2016;133(2):139-146. doi:10.1161/CIRCULATIONAHA.115.018042
- Raitakari O, Kartiosuo N, Pahkala K, et al. Lipoprotein(a) in Youth and Prediction of Major Cardiovascular Outcomes in Adulthood. Circulation. 2023;147(1):23-31. doi:10.1161/CIRCULATIONAHA.122.060667
- Kit BK, Carroll MD, Lacher DA, Sorlie PD, DeJesus JM, Ogden C. Trends in serum lipids among US youths aged 6 to 19 years, 1988-2010. JAMA. 2012;308(6):591-600. doi:10.1001/jama.2012.9136
- Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents; National Heart, Lung, and Blood Institute. Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: summary report. Pediatrics. 2011;128 Suppl 5(Suppl 5):S213-S256. doi:10.1542/peds.2009-2107C
- Ning H, Labarthe DR, Shay CM, et al. Status of cardiovascular health in US children up to 11 years of age: the National Health and Nutrition Examination Surveys 2003-2010. Circ Cardiovasc Qual Outcomes. 2015;8(2):164-171. doi:10.1161/CIRCOUTCOMES.114.001274
- Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2017;38(32):2459-2472. doi:10.1093/eurheartj/ehx144
- Ericsson S, Eriksson M, Vitols S, Einarsson K, Berglund L, Angelin B. Influence of age on the metabolism of plasma low density lipoproteins in healthy males. J Clin Invest. 1991;87(2):591-596. doi:10.1172/JCI115034
- Rudling M, Parini P, Angelin B. Growth hormone and bile acid synthesis. Key role for the activity of hepatic microsomal cholesterol 7alpha-hydroxylase in the rat. J Clin Invest. 1997;99(9):2239-2245. doi:10.1172/JCI119398
- Lea AJ, Clark AG, Dahl AW, et al. Applying an evolutionary mismatch framework to understand disease susceptibility. PLoS Biol. 2023;21(9):e3002311. Published 2023 Sep 11. doi:10.1371/journal.pbio.3002311
- Brassington L, Arner AM, Watowich MM, et al. Integrating the Thrifty Genotype and Evolutionary Mismatch Hypotheses to understand variation in cardiometabolic disease risk. Evol Med Public Health. 2024;12(1):214-226. Published 2024 Jul 31. doi:10.1093/emph/eoae014
- Bietz A, Zhu H, Xue M, Xu C. Cholesterol Metabolism in T Cells. Front Immunol. 2017;8:1664. Published 2017 Nov 27. doi:10.3389/fimmu.2017.01664
- Meilhac O, Tanaka S, Couret D. High-Density Lipoproteins Are Bug Scavengers. Biomolecules. 2020;10(4):598. Published 2020 Apr 12. doi:10.3390/biom10040598
- Gomez-Sanchez CE, Gomez-Sanchez EP. Cholesterol Availability and Adrenal Steroidogenesis. Endocrinology. 2024;165(4):bqae032. doi:10.1210/endocr/bqae032
Deep Dive
The Early Developmental Origins of Atherosclerosis: Lipid Trajectories, Pathobiological Determinants, and the Lifelong Burden of Cardiovascular Risk from Infancy
Introduction
The understanding of atherosclerotic cardiovascular disease (ASCVD) has undergone a profound transformation over the last century, shifting from a perceived inevitable consequence of aging to a recognized life-course metabolic progression that commences during fetal development and infancy.[1] While the clinical manifestations of disease, such as myocardial infarction, stroke, and peripheral vascular syndromes, typically emerge in middle or late adulthood, the silent incubation of these conditions spans decades.[2–4] This developmental paradigm is supported by longitudinal epidemiological cohorts and post-mortem investigations, which collectively indicate that the foundations of cardiovascular pathology are established in the earliest stages of human existence.[2–5] So do babies need statins? Let’s see!
Physiological Trajectories of Plasma Lipids in the First Two Years of Life
The neonatal period and early infancy represent a phase of dramatic metabolic transition characterized by rapid physiological adaptation to extrauterine life. At birth, cholesterol concentrations in umbilical cord blood are substantially lower than those observed in adult populations.[6] However, these levels are highly dynamic and follow distinct trajectories during the first 24 months of life.
Stepwise Increases and Achievement of Early Baseline Levels
Concentrations of atherogenic lipid traits—including low-density lipoprotein cholesterol (LDL-C), non-HDL-C, apolipoprotein B, and lipoprotein(a)—exhibit a consistent stepwise increase from birth through the first 16 months of life.[6] Data from the Copenhagen Baby Heart Study, incorporating 13,354 cord blood samples with longitudinal venous follow-up, demonstrate that by 14–16 months, total cholesterol and LDL-C concentrations approach steady-state levels similar to those observed in adulthood.[1,6]
This observation suggests that the baseline lipid phenotype may be established far earlier than previously hypothesized, constituting a neglected resource for early cardiovascular risk identification.[1]
Variability in Non-Atherogenic Traits
Not all lipid parameters follow a linear rise. Total cholesterol and triglycerides increase sharply in early infancy and subsequently stabilize, whereas HDL-C may rise initially and decline modestly during dietary transition from lipid-rich milk to mixed diets.[6] This variability reflects the metabolic plasticity of infancy as nutritional substrates shift.
Normal Reference Values for Infants and Children
Because lipid levels change rapidly in early life, interpretation requires age-specific reference ranges.[6,7]
Neonatal and Infantile Lipid Ranges (0–24 Months)
Lipid levels at birth are markedly lower than adult thresholds but rise rapidly during infancy, reaching near–adult steady-state concentrations by 14–16 months.[1,6]
Table 1. Neonatal and Infant Lipid Reference Values (Copenhagen Baby Heart Study)[6]
| Age Group | Total Cholesterol (mg/dL) Median (IQR) | LDL-C (mg/dL) Median (IQR) | HDL-C (mg/dL) Median (IQR) | Triglycerides (mg/dL) Median (IQR) |
| Neonates (birth) | 77 (70–93) | 32 (24–39) | 33 (27–41) | 28 (24–34) |
| Infancy (2 months) | 147 (128–166) | 65 (52–79) | 50 (41–62) | 62 (48–84) |
| Early toddler (14–16 months) | 143 (124–162) | 84 (70–103) | 36 (30–45) | 43 (30–60) |
Note: Values reported in mmol/L in Nielsen et al. and converted to mg/dL.[6]
Pediatric Lipid Classification (Ages 2–19 Years)
After age 2 years, lipid interpretation shifts from developmental physiology to cardiovascular risk thresholds. NHLBI pediatric guidelines define the following categories.[7]
Table 2. Pediatric Lipid Classification Thresholds (2–19 Years)[7]
| Classification | Total Cholesterol (mg/dL) | LDL-C (mg/dL) | HDL-C (mg/dL) | Triglycerides (mg/dL) |
| Acceptable | <170 | <110 | >45 | <75 (0–9y) / <90 (10–19y) |
| Borderline | 170–199 | 110–129 | 40–45 | 75–99 (0–9y) / 90–129 (10–19y) |
| High/Abnormal | ≥200 | ≥130 | <40 | ≥100 (0–9y) / ≥130 (10–19y) |
Biological and Environmental Determinants of Infantile Lipid Levels
High cholesterol in infancy reflects a synthesis of biological maturity, genetic inheritance, and early environmental exposure.
Biological Sex and Maternal Health
Female infants exhibit higher total cholesterol, LDL-C, and apoB concentrations than males, differences detectable from birth and persisting through early childhood.[1] Maternal hypercholesterolemia during pregnancy is associated with accelerated early atherogenesis in offspring.[8] Epigenetic analyses of fetal vascular tissue show altered epigenetic regulation of cholesterol-homeostasis pathways (including regulatory regions of SREBP2) in association with early fetal atherosclerotic lesions, supporting fetal metabolic imprinting as a mechanistic framework.[9]
The Nutritional Paradox of Breastfeeding
Breastfed infants often show higher cholesterol concentrations than formula-fed peers, a physiologic response to the cholesterol-rich composition of human milk.[6] Early-life lipid exposure is consistent with lipid tracking across the life course.[10]
Atherosclerosis in Infancy and Youth: Pathological Evidence
Atherosclerosis begins as fatty streaks—subintimal accumulations of foam cells and lymphocytes—that can be identified early in life.[2–4] Autopsy studies demonstrate that early lesions are detectable in infancy and become increasingly prevalent through childhood.[4,11,12]
Pathobiological Determinants of Atherosclerosis in Youth (PDAY)
The PDAY study remains foundational in pediatric cardiovascular pathology. PDAY collected arterial specimens from 2,876 individuals aged 15–34 years who died of external causes.[2] Coronary and aortic lesions were present in adolescence, and lesion severity correlated strongly with modifiable risk factors.[2–4]
Risk scoring derived from PDAY predicts later coronary artery calcification in adulthood, validating the clinical relevance of early-life risk burden.[13]
Historical Trends in Pediatric Lipids
Mean total cholesterol levels among US youths declined between 1988 and 2010, with reductions in both mean levels and prevalence of elevated total cholesterol documented in NHANES analyses.[14] However, these improvements are counterbalanced by the obesity epidemic, which amplifies adverse lipid phenotypes in vulnerable subgroups.[7]
Mechanisms of Age-Related Cholesterol Elevation
Plasma cholesterol rises with aging largely due to reduced fractional clearance of LDL particles.[15] Aging is associated with declining hepatic LDL receptor activity and altered endocrine regulation. Growth hormone influences bile acid synthesis through cholesterol 7α-hydroxylase activity, providing mechanistic links between senescence and dyslipidemia.[16]
The Evolutionary Heritage of High Plasma Cholesterol
High circulating cholesterol, while harmful in modern environments of caloric abundance, may have conferred survival advantages in ancestral contexts characterized by nutritional scarcity and infectious burden.[17]
Evolutionary Advantages
Energy Security. Genetic variants favoring higher circulating cholesterol may have supported energy storage and reproductive fitness during feast-famine cycles.[18]
Immune Modulation. Membrane cholesterol organizes lipid rafts that cluster antigen receptors on memory T cells, enhancing immune responsiveness.[19] HDL also binds and neutralizes bacterial toxins such as lipopolysaccharides during infection.[20]
Endocrine Readiness. Cholesterol is the obligate precursor for steroid hormone synthesis, including cortisol, and is essential for adrenal stress adaptation.[21]
Antagonistic Pleiotropy. Traits conferring early-life survival benefit may persist despite late-life cardiovascular costs.[17]
The Lifelong Burden of Cumulative LDL Exposure
Cardiovascular risk reflects cumulative arterial exposure to atherogenic lipoproteins rather than single time-point measurements.[1,22] Genetic, epidemiologic, and clinical evidence establishes LDL-C as a causal driver of ASCVD, emphasizing the importance of early prevention.[22]
Clinical Management and the Window of Opportunity
Universal lifestyle promotion and targeted screening are central to prevention. NHLBI guidelines recommend universal lipid screening at ages 9–11 years and earlier screening beginning at age 2 in high-risk families.[7]
Universal Lifestyle Interventions
The Cardiovascular Health Integrated Lifestyle Diet (CHILD-1) provides the foundation of primordial prevention beginning in early childhood.[7]
- Exclusive breastfeeding when possible[6]
- Delaying solid foods until at least 4 months[7]
- Transition to reduced-fat dairy after 12 months in appropriate contexts[7]
- Total fat intake of ~25–30% of calories after age 2 with saturated fat limitation[7]
Pharmacologic therapy is reserved for older children with severe dyslipidemia, particularly familial hypercholesterolemia.[7]
Conclusion
Atherosclerosis is a lifelong process beginning in fetal development and infancy. Early lipid trajectories establish cumulative exposure burdens that drive lesion progression from fatty streaks in childhood to fibrous plaques in adulthood.[2–4] Prevention must therefore begin early—optimizing maternal health, identifying high-risk lipid phenotypes in infancy, and implementing primordial prevention during the developmental window when vascular injury remains minimal and modifiable.[1,7,8] Infants are not eligible for statin therapy at this time but there certainly are things the mother can do when pregnant and after birth to protect her child for years to come.
References
- Holven KB. Plasma cholesterol levels in infancy: a neglected resource. Eur Heart J. 2023;44(42):4419-4421. doi:10.1093/eurheartj/ehad558
- Natural history of aortic and coronary atherosclerotic lesions in youth. Findings from the PDAY Study. Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group. Arterioscler Thromb. 1993;13(9):1291-1298. doi:10.1161/01.atv.13.9.1291
- McGill HC Jr, McMahan CA, Zieske AW, et al. Association of Coronary Heart Disease Risk Factors with microscopic qualities of coronary atherosclerosis in youth. Circulation. 2000;102(4):374-379. doi:10.1161/01.cir.102.4.374
- Strong JP, Malcom GT, McMahan CA, et al. Prevalence and extent of atherosclerosis in adolescents and young adults: implications for prevention from the Pathobiological Determinants of Atherosclerosis in Youth Study. JAMA. 1999;281(8):727-735. doi:10.1001/jama.281.8.727
- Luca AC, David SG, David AG, et al. Atherosclerosis from Newborn to Adult-Epidemiology, Pathological Aspects, and Risk Factors. Life (Basel). 2023;13(10):2056. Published 2023 Oct 14. doi:10.3390/life13102056
- Taageby Nielsen S, Mohr Lytsen R, Strandkjær N, et al. Significance of lipids, lipoproteins, and apolipoproteins during the first 14-16 months of life. Eur Heart J. 2023;44(42):4408-4418. doi:10.1093/eurheartj/ehad547
- Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents; National Heart, Lung, and Blood Institute. Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: summary report. Pediatrics. 2011;128 Suppl 5(Suppl 5):S213-S256. doi:10.1542/peds.2009-2107C
- Napoli C, Glass CK, Witztum JL, Deutsch R, D’Armiento FP, Palinski W. Influence of maternal hypercholesterolaemia during pregnancy on progression of early atherosclerotic lesions in childhood: Fate of Early Lesions in Children (FELIC) study. Lancet. 1999;354(9186):1234-1241. doi:10.1016/S0140-6736(99)02131-5
- de Nigris F, Cacciatore F, Mancini FP, et al. Epigenetic Hallmarks of Fetal Early Atherosclerotic Lesions in Humans. JAMA Cardiol. 2018;3(12):1184-1191. doi:10.1001/jamacardio.2018.3546
- Øyri LKL, Bogsrud MP, Kristiansen AL, et al. Cholesterol at ages 6, 12 and 24 months: Tracking and associations with diet and maternal cholesterol in the Infant Cholesterol Study. Atherosclerosis. 2021;326:11-16. doi:10.1016/j.atherosclerosis.2021.04.017
- Tanaka K, Masuda J, Imamura T, et al. A nation-wide study of atherosclerosis in infants, children and young adults in Japan. Atherosclerosis. 1988;72(2-3):143-156. doi:10.1016/0021-9150(88)90075-5
- Françoso LA, Coates V. Anatomicopathological evidence of the beginning of atherosclerosis in infancy and adolescence. Arq Bras Cardiol. 2002;78(1):131-142.
- Gidding SS, Rana JS, Prendergast C, et al. Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Risk Score in Young Adults Predicts Coronary Artery and Abdominal Aorta Calcium in Middle Age: The CARDIA Study. Circulation. 2016;133(2):139-146. doi:10.1161/CIRCULATIONAHA.115.018042
- Kit BK, Carroll MD, Lacher DA, Sorlie PD, DeJesus JM, Ogden C. Trends in serum lipids among US youths aged 6 to 19 years, 1988-2010. JAMA. 2012;308(6):591-600. doi:10.1001/jama.2012.9136
- Ericsson S, Eriksson M, Vitols S, Einarsson K, Berglund L, Angelin B. Influence of age on the metabolism of plasma low density lipoproteins in healthy males. J Clin Invest. 1991;87(2):591-596. doi:10.1172/JCI115034
- Rudling M, Parini P, Angelin B. Growth hormone and bile acid synthesis. Key role for the activity of hepatic microsomal cholesterol 7alpha-hydroxylase in the rat. J Clin Invest. 1997;99(9):2239-2245. doi:10.1172/JCI119398
- Lea AJ, Clark AG, Dahl AW, et al. Applying an evolutionary mismatch framework to understand disease susceptibility. PLoS Biol. 2023;21(9):e3002311. Published 2023 Sep 11. doi:10.1371/journal.pbio.3002311
- Brassington L, Arner AM, Watowich MM, et al. Integrating the Thrifty Genotype and Evolutionary Mismatch Hypotheses to understand variation in cardiometabolic disease risk. Evol Med Public Health. 2024;12(1):214-226. Published 2024 Jul 31. doi:10.1093/emph/eoae014
- Bietz A, Zhu H, Xue M, Xu C. Cholesterol Metabolism in T Cells. Front Immunol. 2017;8:1664. Published 2017 Nov 27. doi:10.3389/fimmu.2017.01664
- Meilhac O, Tanaka S, Couret D. High-Density Lipoproteins Are Bug Scavengers. Biomolecules. 2020;10(4):598. Published 2020 Apr 12. doi:10.3390/biom10040598
- Gomez-Sanchez CE, Gomez-Sanchez EP. Cholesterol Availability and Adrenal Steroidogenesis. Endocrinology. 2024;165(4):bqae032. doi:10.1210/endocr/bqae032
- Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2017;38(32):2459-2472. doi:10.1093/eurheartj/ehx144